
An international scientific team, including CONICET researcher Daniel Alonso, director of the Center for Molecular and Translational Oncology (COMTra-UNQ), has just released the first Mouse Cancer Cell line Atlas. This valuable tool, useful for understanding how different types of cancer develop and can be treated in humans, has as its first fruit a work published in the prestigious journal Nature, which analyzes the specific relationships between the KRAS gene (which appears mutated in many types of cancer) and the development of tumors in different organs.
Mouse models of cancer and other diseases have become indispensable resources and a source of key discoveries for understanding their mechanisms of action, as well as for the development of new treatments.
**“Until now, although there were collections of human cell lines, there was no resource of these characteristics for cancer in mice, which is the most widely used experimental model in biomedical research and the one that best reproduces the disease in humans,” said Alonso.
The development of the Atlas was led from Germany by scientists Sebastian Mueller and Roland Rad, from the Munich School of Medicine and Health and the Heidelberg Cancer Research Center, who gathered the collection of cell lines by convening researchers from other centers in Germany, the United Kingdom, Canada, the United States, Spain, Italy and Argentina.
“My contribution included a cell line of an aggressive cancer, which we isolated more than three decades ago during my doctoral thesis together with researcher Elisa Bal at the Roffo Oncology Institute (UBA) and validated as a model during all these years at the National University of Quilmes (UNQ). I serve as the Latin American contact for this international initiative,” *Alonso said.
The KRAS oncogene in different cancers #
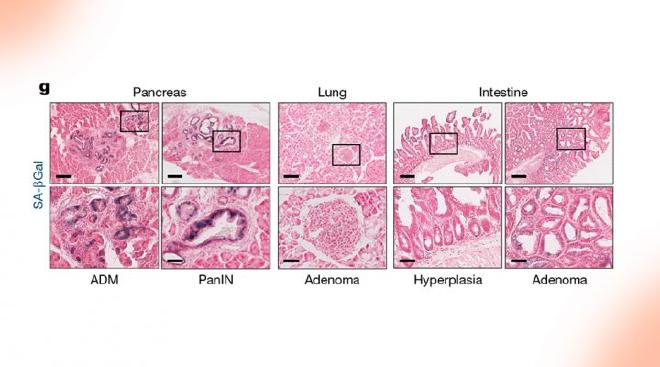
In the study recently published in Nature (of which Alonso is also one of the authors), the atlas was used as a tool to study the specific evolution of cancers in each tissue under the impulse of the KRAS oncogene.
“Oncogenes such as KRAS show great specificity depending on the tissue in which they act; Its ability to cause cancer, genetic interactions, and effects on cells vary greatly, but the reasons behind this are still not entirely clear. In some ways, the precise links between the mutated oncogene and cancer progression, to the complex changes in cellular behavior that lead to cancer in one tissue, but not in others, constitute a ‘missing link’ to better address prevention and treatment,” Alonso said.
The study made it possible to analyse these questions in types of cancer strongly associated with the KRAS oncogene, such as pancreatic, lung and intestinal cancer. “In addition, as these evolutionary processes begin to be understood in these mouse cell lines, the model studied itself is a ’test bed’ to advance towards new therapeutic approaches for human cancer. In fact, part of the study sought the parallelism of the results with clinical data,” added the CONICET researcher.
“More generally, these findings begin to shed light on the innate and acquired factors that guide the evolution of cancer in different tissues, and may eventually establish more predictable molecular patterns and temporal dynamics, with expected results in the cell compartment. It is an important advance towards a mechanistic understanding of the always complex and unstable cancerous genomes,” concluded Alonso.
Citation #
- The article A disease model resource reveals core principles of tissue-specific cancer evolution was published in Nature magazine. Authors: Sebastian Mueller, Niklas de Andrade Krätzig, Markus Tschurtschenthaler, Miguel G. Silva, Chiara Thordsen, Riccardo Trozzo, Perrine Simon, Frederic Saab, Thorsten Kaltenbacher, Magdalena Zukowska, Daniele Lucarelli, Rupert Öllinger, Joscha Griger, Nina Groß, Tanja Groll, Jessica Löprich, Antonio E. Zaurito, Linus R. Schömig, Jeroen M. Bugter, Stefanie Bärthel, Chiara Falcomatà, Alexander Strong, Cordelia Brandt, Mulham Najajreh, Aristeidis Papargyriou, Roman Maresch, Katharina A. N. Collins, David Sailer, Christian Schneeweis, Sebastian Burger, Lisa M. Fröhlich, Christine Klement, Alexander Belka, Juan J. Montero, Ute Jungwirth, Maximilian Reichert, Markus Moser, Jens Neumann, George Vassiliou, Juan Cadiñanos, Ignacio Varela, Carsten Marr, Daniel F. Alonso, Pier-Luigi Lollini, Jean Zhao, Louis Chesler, Clare M. Isacke, Angela Riedel, Christian J. Braun, Martin L. Sos, Filippo Beleggia, Hans C. Reinhardt, Monica Musteanu, Mariano Barbacid, Michael Quante, Marc Schmidt-Supprian, Günter Schneider, Simon Clare, Trevor D. Lawley, Gordon Dougan, Katja Steiger, Nathalie Conte, Allan Bradley, Lena Rad, Dieter Saur & Roland Rad
Mueller, S., de Andrade Krätzig, N., Tschurtschenthaler, M. et al. A disease model resource reveals core principles of tissue-specific cancer evolution. Nature (2026). https://doi.org/10.1038/s41586-026-10187-2
- The article Elaboran el primer atlas de líneas celulares de cáncer de ratón signed by Miguel Faigón, was published today on CONICET’s website
Contact [Notaspampeanas](mailto: notaspampeanas@gmail.com)

